
How does the electronic configuration of an atom relate to its position in the Modern Periodic Table ?Ĭompare and contrast the arrangement of elements in Mendeleev’s Periodic Table and the Modern Periodic Table. Which of these will be more electronegative ? Why ? Write the electronic configuration of these two elements. Nitrogen (atomic number 7) and phosphorus (atomic number 15) belong to group 15 of the periodic table. (d) Which type of ion, cation or anion, will be formed by element A ? (c) Will C be larger or smaller in size than B ? (b) State whether C is more reactive or less reactive than A. (a) State whether A is a metal or non-metal. The positions of three elements A, B and C in the periodic table are shown below : In the Modern Periodic Table, calcium (atomic number 20) is surrounded by elements with atomic numbers 12,19,21 and 38. What is the atomic mass of calcium Atomic Mass of Calcium. There are many isotopes of calcium, ranging from calcium-40 with 20 neutrons, to calcium-48 with 28 neutrons. Atoms of the same element with different numbers of neutrons are isotopes. (b) To which of the following elements would it be chemically similar ? (Atomic numbers are given in parentheses.) However, the number of neutrons in the nuclei of the calcium atoms can vary. 
(a) What is the atomic number of this element ? This means that every atom of calcium contains 20 protons. as fluorine have in common ?Īn atom has electronic configuration 2, 8, 7. According to the Periodic Table of Elements, calciums atomic number is 20. (b) What property do all elements in the same column of the Periodic Table. (a)What property do all elements in the same column of the Periodic Table as boron have in common ? There are many isotopes of calcium, ranging from calcium-40 with 20 neutrons, to calcium-48 with 28. However, the number of neutrons in the nuclei of the calcium atoms can vary. (e)twice as many electrons in its second shell as in its first shell ? Calcium's atomic number is 20, which means all calcium atoms have 20 protons in their nuclei. (d) a total of two shells with three electrons in its valence shell. (c) a total of three shells, with four electrons in its valence shell ? (b) the electronic configuration 2, 8, 2 ? (a) two shells, both of which are completely filled with electrons ? X would most likely to be in the same group of the periodic table as (c)The atoms lose their electrons more easily.Įlement X forms a chloride with the formula \( XCl_2, \) which is solid with a high melting point. (b) The number of valence electrons increases. (a) The elements become less metallic in nature. Isotopes Atoms of the same element with different numbers of neutrons. Where more than one isotope exists, the value given is the abundance weighted average. The alkaline earth metals are six chemical elements in group 2 of the periodic table.They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). This is approximately the sum of the number of protons and neutrons in the nucleus.  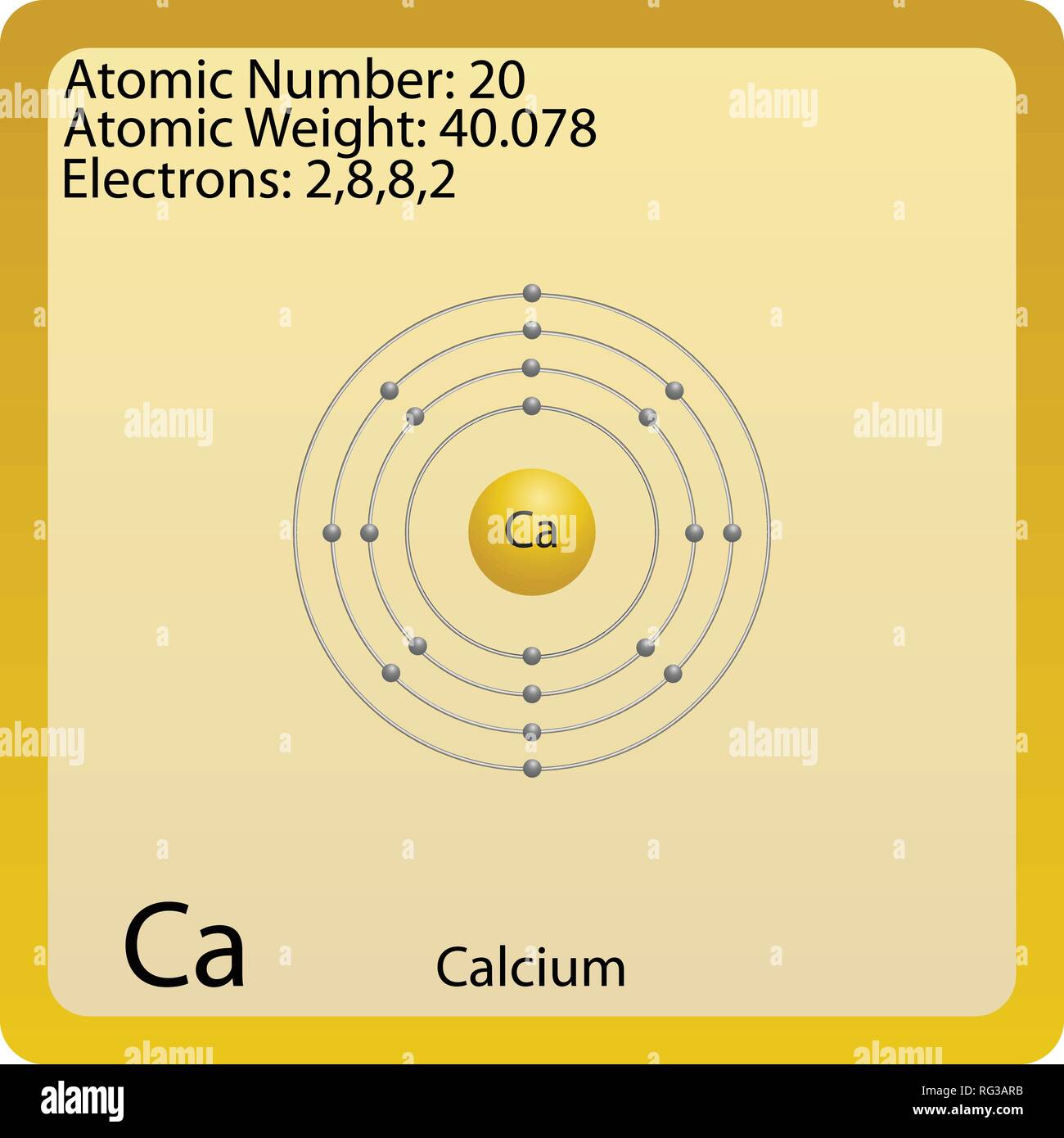
Simply use this information to obtain its electronic configuration. Which of the following statements is not a correct statement about the trends wlien going from left to right across the periods of Periodic Table. It provides atomic mass, mass excess, nuclear binding energy, nucleon separation energies, Q-values, and nucleon residual interaction parameters for atomic. Relative atomic mass The mass of an atom relative to that of carbon-12. Ca: Ar 4s2 Calcium has an atomic number of 20. With atomic number 88, it has four natural isotopes of atomic weight 228, 226, 224 and 223 - though there are a remarkable 21 more artificial isotopes. You will need to refer to a periodic table for proton values.NCERT solutions of related questions for Periodic Classification of Elements It's classified in the periodic table as an alkaline earth metal - the heaviest of the series - putting it alongside more familiar metals like magnesium and calcium. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Calcium (Ca) is a silvery-white soft metal that has the atomic number 20 in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
